A 4-hour-old thoroughbred colt foal presented to the hospital following an assisted vaginal delivery at term. The mare's placenta was reported to be normal.
The foal exhibited abnormal activity after birth, was unable to stand or maintain sternal recumbency with poor suckle reflex. These signs were thought to be consistent with neonatal maladjustment syndrome. A nasogastric tube was passed and colostrum milked from the mare was given. A dose of tetanus anti-toxin was administered, and the foal was referred for further evaluation and supportive treatment.
Clinical examination
On admission the foal was responsive, non-ambulatory and was carried to the stable. A holosystolic heart murmur was auscultated, with a heart rate of 116 beats per minute (bpm); (reference range 80–120 bpm) (all reference ranges from Paradis, 2006).
Mucous membranes were pink but slightly injected with capillary refill time greater than 2. The foal respiratory rate was increased (60 breaths per minute; reference range: 20–40 breaths per minute); the breathing was slightly laboured, and the foal was hypothermic (36.6˚C; reference range: 37–39˚C).
There were palpable rib fractures on the left hand side of the chest. The foal had flexural limb deformities: moderately contracted carpi and hypoextension of the tarsus. The foal also showed clinical signs consistent with dysmaturity; slightly floppy ears and small body size.
Palpation of the umbilicus and joints were within normal limits.
Investigations
On arrival, full haematology, biochemistry, arterial blood gas and lactate analysis were carried out. A blood culture sample was aseptically obtained to diagnose/rule out sepsis, and assist in appropriate choice of antimicrobial treatment.
The total leukocyte count was within normal limits (9.9 x 109/litre; reference range: 6.2–12.4 x 109/litre). This indicates a reduced risk that the foal's clinical signs were caused by sepsis. The packed cell volume was checked because of the risk of haemothorax from the fractured ribs, and was normal (42 l/l; reference range: 35–43 l/l).
Peripheral lactate was measured to assess tissue perfusion, with hyperlactataemia noted (5.4 mmol/litre; reference range: <2 mmol/litre). The blood biochemistry profile reported a significant degree of azotaemia, urea (7.9 mmol/litre; reference range: 2.8–4.1 mmol/litre), and creatinine (1158 μmol/litre; reference range: 97–138 μmol/litre). Hypoglobulinaemia (47g/litre; reference range: 53–73 g/litre) was recorded.
Arterial blood gas measurements were obtained to check for hypoxaemia, hypercapnia and tissue perfusion, the results identified a degree of hypoxaemia with PO2 65 mmHg (reference range 80–100 mg), hypercapnia 57 mmHg (reference range: 36–46 mmHg) and oxygen saturation (SPO2) being 93% (reference range 94–100%).
Radiography of both carpi and tarsi showed complete ossification of the joints, further radiographs of the thorax were taken, followed by thoracic and abdominal ultrasound. Both imaging techniques confirmed the presence of multiple rib fractures on the left hand side. There was also significant haemothorax, and the rib fractures were in close proximity to the pericardium.
Both eyes were evaluated for fluorescein uptake to rule out corneal trauma from the foal thrashing around in lateral recumbency, but were negative.
Diagnosis
The diagnosis was neonatal maladjustment syndrome, dysmature with fractured ribs, haemothorax, renal compromise and flexural limb deformities.
Treatment
The foal was separated from the mare, placed on a soft padded mattress, another padded mattress was placed against the wall. The purpose of these was to prevent trauma to the foal particularly if the foal was thrashing to try to stand or was seizuring. The heat lamp in the stable was turned on to warm the foal because the foal was hypothermic. Once the foal's temperature was within normal limits, it was switched off.
Oxygen therapy was initiated immediately, an indwelling nasal cannula (Ryles tube) was placed in the left nostril, sutured and taped in place, and humidified oxygen was administered at a flow rate of 6 litres/minute. This was adjusted to 8 litres/minute following the first arterial blood gas results, as there was significant hypoxaemia, hypercapnia and low oxygen saturation (SPO2).
An intravenous polyurethane over the wire catheter (Mila International) was inserted in the left jugular vein aseptically. Crystalloid fluid therapy was started with a 1 litre bolus of Hartmanns solution on admission, followed by a 5% glucose isotonic solution at 3 ml/kg/hour. Two litres of frozen plasma (Hypermune®, Veterinary Immunogenics) were administered at 2 ml/kg/hour due to hypoglobulinaemia, and partial failure of passive transfer.
A nasogastric tube was placed in the right nostril and a radiograph was taken to confirm it was in the correct position. The mare was milked every 2 hours to feed the foal, the foal was refluxed prior to each feed using a dosing syringe, and aspirated gently to check for reflux. Then 150 mls of the mare's milk was fed every 2 hours by gravity flow via the nasogastric tube, with the foal standing or in sternal recumbency. The amount was increased slowly over the next 24 hours to 500 ml, and the foal's tolerance and comfort levels were assessed.
Medication was given as detailed in Table 1
Table 1. Medications administered within the first 24 hours
| Allopurinol (40 mg/kg) PO once off |
| Ceftiofur (5–10 mg/kg) IV QID |
| Tranexamic acid (20 mg/kg) BID IV q 3 days |
| Selenium (2 ml/50 kg) IM once off treatment |
| Meloxicam (0.6 mg/kg) IV BID |
| Buprenorphine (0.3 mg/10 kg) IV TID |
| Chloramphenicol ointment was applied to both eyes q 4 hours. |
| Vitamin C (20–100 mg/kg) PO SID |
| Vitamin E (10 iu(mg)/kg) PO SID |
PO = per os, IV = intravenous, IM = intramuscular, SID = once daily, BID = twice daily, TID = three times daily, QID = four times daily, q = every
Meconium was not seen to be passed and the foal was seen to be straining. A fleet enema was administered, and the foal successfully passed a large amount of meconium.
An indwelling urinary catheter was inserted aseptically, and connected to a closed collection system to allow accurate monitoring of urine output and maintenance of a clean bed.
The umbilicus was dipped in a 0.5% chlorhexidine solution, and this treatment continued every 6 hours for 48 hours.
The treatment/nursing plan was as following and is outlined in Figure 1.
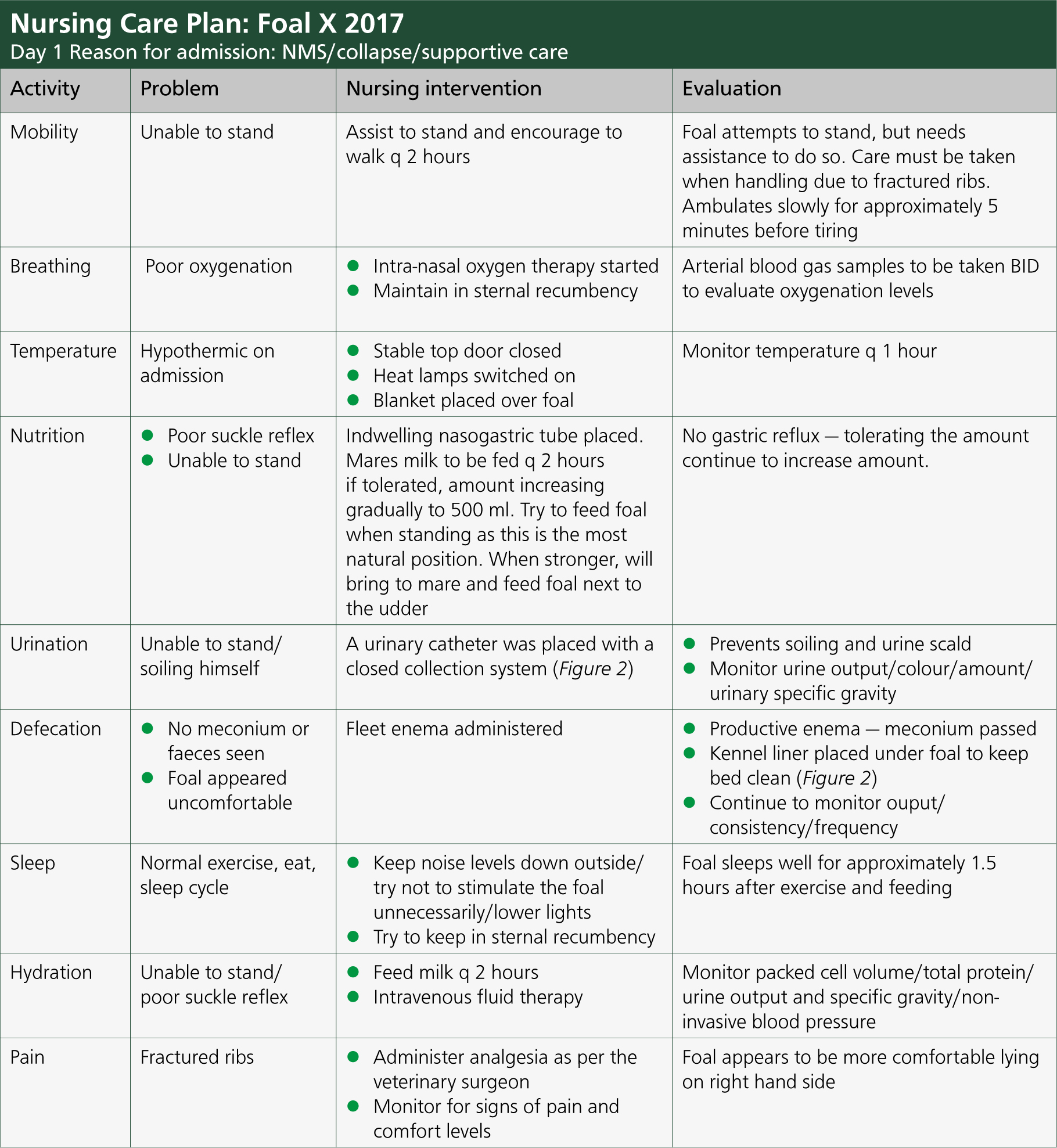

The treatment/nursing plan
Mare
A full clinical examination was performed on the mare on admission. The mare was milked every 2 hours and a clinical examination performed twice daily, as per the clinician's instructions. Oxytocin was administered 10 IU intramuscularly (IM) twice daily to facilitate involution of the uterus.
Foal
A full clinical examination was performed, and the foal was helped to stand and turned every 2 hours. It was important to change the foal's position every 2 hours to aerate each lung (weak/dummy foals tend to slip into lateral recumbency) (Figure 3). This could also help prevent the development of decubitus ulcers on bony prominences. Arterial blood gas sampling and lactate were measured twice daily. Non-invasive blood pressure (NIBP) was measured every 2 hours using a tail cuff — mean arterial pressure (MAP)(reference range: MAP <65–>85).

Case progression
The foal responded positively to treatment, but the progress was slow. Within the first 24 hours the foal was able to stand with assistance (two people) (Figure 4), and slowly walked around the box for 5 minutes before tiring, assisted again to lie down and positioned in sternal recumbency. The position of the fractures increased the chance of the ribs puncturing the lung or heart.
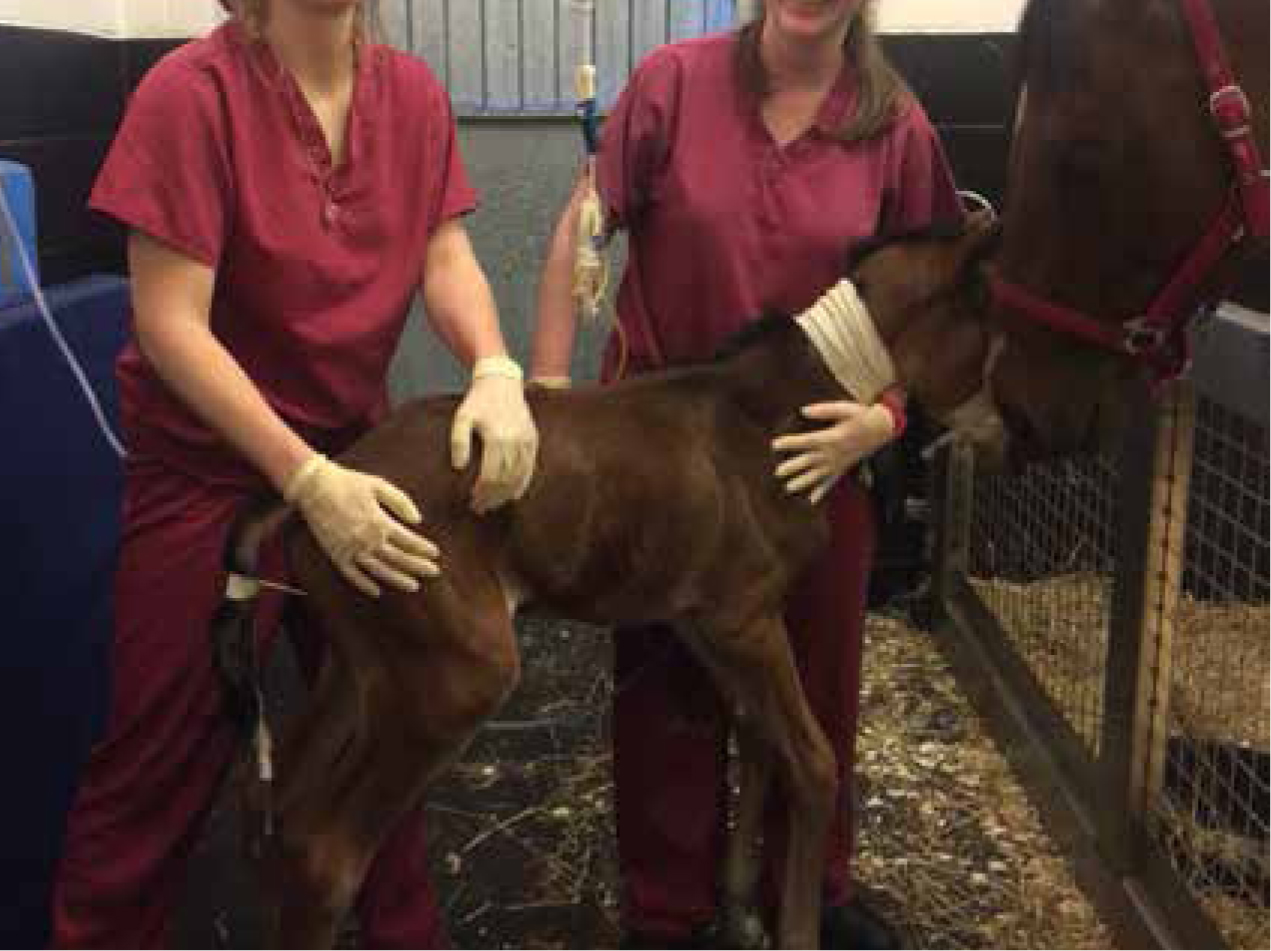
The treatment plan remained the same for the following 5 days with the foal standing/walking for longer periods. The foal was brought into the mare's section every 2 hours to encourage maternal interaction. The foal was fed via the nasogastric tube while standing, with efforts being made to feed when standing next to the mare's udder. This was done to promote normal behaviour. This procedure was repeated every 2 hours. The foal showed signs of improvement in suckle reflex and interest in the udder. On Day 2 the foal latched on to the udder and nursed successfully for over a minute. Repeated haematology and biochemistry reported the packed cell volume (PCV) remaining within the normal reference range, and the renal parameters were within normal limits after 2 days of fluid therapy.
After 2 weeks the foal was getting up and down by himself and nursing well.
The haemothorax and the stability of the rib fractures were closely monitored by ultrasonography over the subsequent weeks. The foal was treated conservatively with strict box rest. Once signs of callus formation and stability of the rib fractures was improved, a small amount of controlled turnout was started. The foal was hospitalised for 1 month in total and was reported to be doing well at 6 months.
Discussion
This case report details the intensive care of a neonatal foal with moderate neonatal maladjustment syndrome, some signs of dysmaturity, concurrent failure of passive transfer and rib fractures that had resulted in a haemothorax. Neonatal maladjustment syndrome is a disorder of the newborn foal linked with variable degrees of hypoxia before or after parturition.
The foal was reported as suffering a prolonged delivery with force been required, which means that the foal was at a high risk for the development of neonatal maladjustment syndrome (Haggett, 2013). Dysmaturity is defined as a foal born to normal term (320 days or more), but which exhibits the physical characteristics of prematurity, for example silky fine coat, small size, floppy ears and incomplete ossification of joints (Carr, 2014).
A holosystolic heart murmur was auscultated on the foal's initial clinical examination; this type of murmur is commonly heard in foals under the age of 7 days due to the delayed closure of ductus arterious (Haggett, 2013), and generally resolves within 3–4 days.
Initial blood biochemistry results reported hypoglobulinaemia and this is a crude indictor of failure of passive transfer. Failure of passive transfer occurs when a foal fails to ingest or absorb adequate colostral immunoglobulins (McAuliffe and Slovis, 2008). Foals are born with a naïve immune system, and rely on the maternal antibodies present in the colostrum for protection from infectious diseases in the first few weeks of life. Many factors lead to an increased risk of failure of passive transfer, but one of the main reasons in this case was the foal's inability to stand and nurse. Despite the foal receiving some colostrum via nasogastric tube prior to travel, he was recumbent in the trailer for the 5 hour travel time, and therefore not receiving maternal milk during this time. In addition, initial blood biochemistry results reported hypoglobulinaemia, and this is a crude indicator of failure of passive transfer. As a result of the hypoglobulinaemia and clinical history of the foal, a plasma transfusion was administered, as 1 litre of commercial plasma can increase the IgG concentration by 2 g/litre (Dunkel, 2014).
Creatinine was significantly increased on the foal's initial biochemistry (1158 μmol/litre; reference range: 97–138 μmol/litre), but this is often seen in foals less than 24 hours old that have had some degree of placental insufficiency, birth hypoxia or maladjustment (Haggett, 2013). Blood test results 48 hours later showed the creatinine level had returned to within normal limits following fluid therapy.
Following the ophthalmic examination no abnormalities were detected; prophylactic antibiotic ointment (chloramphenicol) was started as the foal was recumbent, so there was an increased risk of corneal trauma. There is evidence that corneal sensitivity in foals is very poor. Foals also have lower tear production compared with that of adults, which can lead to an increased incidence of corneal disease in sick neonatal foals (McAuliffe and Slovis, 2008).
A nasal cannula was placed in the left nostril and the foal received oxygen therapy for 5 days. The nasal cannula was replaced daily as they can become blocked with nasal secretions or can kink (Stoneham, 2012). It was important to maintain the foal in sternal recumbency as lateral recumbency causes poorer ventilation of the lower lung; also in this case the foal seemed uncomfortable lying on his left hand side, most likely due to the fractured rib, and as a result the foal was more restless when lying on this side. The foal was turned every 2 hours to prevent pressure sores (Galvin et al, 2004).
Intravenous catheter care is very important in the sick, compromised neonate. Polyurthehane over the wire catheters were chosen in this case, as they are the least thrombogenic, but need experience to place. The intravenous catheter should be assessed at least twice daily for heat, swelling and discharge, and patency confirmed via flushing with heparinised saline every 6 hours. Thrombophlebitis and loss of catheter patency can lead to patient discomfort and the need for a replacement intravenous catheter (Rippingale and Fisk, 2013). A light covering, for example some bandaging tubigrip, was placed over the neck to help protect and cover the intravenous catheter.
A 5% glucose solution was chosen for the neonatal foal; glycogen stores present at birth are sufficient for only approximately 2 hours of the energy requirements in the unfed foal, and fat stores are also very low at birth (Corley et al, 2008). In addition, foals have limited ability to excrete excess sodium, so a Hartmanns solution is not recommended for maintenance (Haggett, 2013).
A plasma transfusion should be treated like a blood transfusion, the transfusion is administered slowly at first with vital parameters recorded every 5 minutes for the first 30 minutes, and the foal monitored for any adverse reactions. Adverse reactions may include tachycardia, tachypnea, pyrexia, oedema, urticaria and signs of anaphylaxis.
Extreme care was taken by the carers when assisting the foal to stand. Two people were required to support the foal until it was strong and stable enough to stand with just some assistance on the tail. Incorrect/rough handling of a foal with rib fractures can have disastrous consequences, resulting in death due to the displaced fracture causing laceration to the heart/greater vessels (Russell et al, 2006). According to a study by Schambourg et al (2003), 89% of foals with fatal rib fractures died within 1 week of birth. In the case described here, there was a discussion with the client regarding surgical treatment of the fractured ribs, but they opted for conservative treatment.
A neonatal foal should receive 10% of its bodyweight in milk each day. The carer must ensure the nasogastric tube is in the correct position before each feeding to help prevent aspiration pneumonia. The patient must be monitored for signs of colic, discomfort and abdominal distension. Ileus is common in foals with neonatal maladjustment syndrome (Haggett, 2013).
Once the foal was permitted short controlled exercise, the foal was weighed daily to monitor progress. In healthy, normal foals, growth rate is rapid during the first month, with Thoroughbred foals gaining 1.5–1.7 kg/day (Paradis, 2006).
The foal's urine output was monitored every 2 hours, and this should be a minimum of 2 ml/kg/hour. The collection bag was emptied as needed. The urine specific gravity (USG) is also a good indicator of hydration; normal hydrated foals usually have a USG below 1.010 (Haggert, 2013)
Arterial blood samples were obtained twice daily in the first 2 days of hospitalisation, the foal reacted to the insertion of the needle increasing the risk of causing a haematoma. Local anaesthetic cream (Emla cream) was applied 5 minutes before obtaining the sample, and this worked very effectively, and was less stressful for the foal.
As mentioned previously, a new study has shown an association between neonatal maladustment syndrome and a persistence of high concentrations of neurosteroid in the foal in the post-natal period (Aleman et al, 2017). These neurosteroids are thought to be partly responsible for keeping the foal in a sleep like state of unconsciousness while in utero. It is thought that physical pressure (squeezing) on the foal while in the birth canal during labour, signals to the foal to stop producing the sedative inducing neurosteroids, and causes the foal to ‘wake up’; during a normal birth, foals experience such a physical compression for about 20 minutes during stage 2 labour. There is a novel procedure called ‘The Madigan foal squeeze procedure’, which is currently been used in the treatment of NMS foals, and success rates are currently being documented (Madigan et al, 2012). The procedure involves a rope been applied to the foal's upper torso to mimic the pressure normally experienced in the birth canal. It is suspected that the pressure triggers biochemical changes in the central nervous system that are critical for transitioning the foal from a sleep-like state in the womb to wakefulness at birth. Research gathered has revealed that some foals that received the squeeze procedure recovered faster than those that received medical therapy only. In this study, foals that received the squeeze procedure with or without medical therapy were 3.7 times more likely to recover faster than foals that did not receive the squeeze procedure (Aleman et al, 2017). Unfortunately this technique was not suitable is this case as the patient had broken ribs.
Conclusion
A lot of hard work and dedication by the entire veterinary team is required in the intensive care of a recumbent neonate, but particularly from the nursing team. It is particularly rewarding to see these patients make a full recovery.
KEY POINTS
- Neonatal maladjustment syndrome (NMS) is not completely understood but is linked with variable degrees of hypoxia before or after parturition.
- Recent studies have shown an association of NMS with a persistence of high concentrations of neurosteroid in the post-natal period.
- The foal was diagnosed — NMS, dysmature with fractured ribs, haemothorax, renal compromise and flexural limb deformities.
- Every 2 hours, a clinical examination of the foal was performed, the foal was a assisted to stand and fed via nasogastric tube.
- The Madigan foal squeeze technique is a new procedure currently been used in the treatment of NMS foals.


